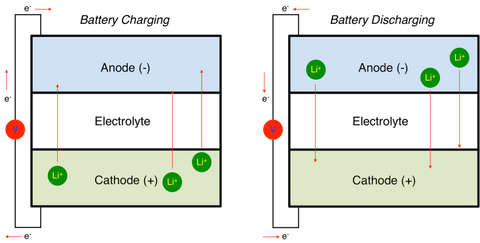
If the battery is used for a long time, the color may be dark. In the case where the "+" and "-" signs are blurred, the red-coated battery pole represents the positive electrode, and the pole marked with the blue mark is the negative electrode. Identification of positive and negative batteries The total reaction is a spontaneous (release energy) redox reaction. Two active conductors are used as electrodes to keep the two electrodes in contact and inserted into the electrolyte solution (loop). General conditions for forming a simple primary battery Negative electrode: Loss of electrons, oxidation reaction, and a pole of electrons flowing from the external circuit.Įxternal circuit: electrons flow from the negative electrode to the positive electrode current flows from the positive electrode to the negative electrode.Ĭircuit inside the ion of Yin and Yang of directional movement: anionic move to the cathode, cationic move to the anode.Ģ, the internal and external circuit current direction and the conductive particles moving directionģ. From a physical point of view, it is a pole of electrons flowing out of the circuit. In the electrolytic cell, the electrode that acts as a reduction is distinguished from the primary battery. In the galvanic cell, it refers to the electrode that acts as an oxidation, and the battery reaction is written on the left side. The negative pole refers to the lower end of the potential (potential) in the power supply. The product contains volatiles such as asphalt, does not contain washing oil, and the washing oil has been pre-treated by the prescription. Process Description Graphite is sintered under a 99.99% nitrogen atmosphere. 
The anode materials being explored are nitrides, PAS, tin-based oxides, tin alloys, nano-anode materials, and other intermetallic compounds.įully automatic atmosphere protection tunnel type resistance furnace is used for the firing of graphite anode material. At present, the anode materials that have been practically used for lithium ion batteries are generally carbon materials such as graphite, soft carbon (such as coke, etc.), hard carbon, and the like. In general, the choice of a good anode material should follow the following principles: higher specific energy lower electrode potential than lithium electrode good reversibility of charge and discharge reaction good compatibility with electrolyte and binder small specific surface area (2.0g/cm3) good size and mechanical stability during lithium insertion abundant resources, low price stable in air, no toxic side effects. Whether the lithium-ion battery can be successfully produced is the key to whether a negative electrode material capable of reversibly deintercalating lithium ions can be prepared. The negative electrode of the lithium ion battery is made by mixing a negative electrode active material carbon material or a non-carbon material, a binder and an additive to form a paste adhesive, which is evenly coated on both sides of the copper foil, and dried and rolled. Currently, the common negative electrode materials include a carbon negative electrode material, a tin-based negative electrode material, a lithium-containing transition metal nitride negative electrode material, an alloy negative electrode material, and a nano-scale negative electrode material. The negative electrode material refers to a raw material constituting the negative electrode in the battery. In primary cell, refers to the oxidation of electrodes, battery reaction in writing on the left. The cathode the potential (voltage) refers to the power lower end. Total reaction: PbO2 + 2 - h2so4 pbso4 + Pb = 2 + 2 h2o The positive: PbO2 + 2 + HSO4 - e + 3 h + = PbSO4 + 2 h2o The lead storage battery is made up of a group of lead-bismuth alloy grids filled with spongy metal lead, and the other group is made of lead-bismuth alloy grid filled with alumina.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
